What We Do
Our Services
and Commitments
From fast-track approval to commercial execution, ACA Pharma helps innovative medicines and devices reach patients faster through Macau, Hong Kong and Singapore pathways, then expands access into Mainland China and other key markets through compliant, end-to-end commercialization.
Learn More
Macau and Hong Kong Fast-Track for Early Market Access
ACA Pharma specializes in fast-track registration and commercialization pathways that can enable market access in as little as 30 to 90 days post-submission. For medicines through Macau and medical devices through Hong Kong, this is the fastest compliant path into Mainland China’s pilot zones — giving partners a faster route to market, earlier patient access, and a strategic advantage in Greater China.

Expanding Access. Extending Reach.
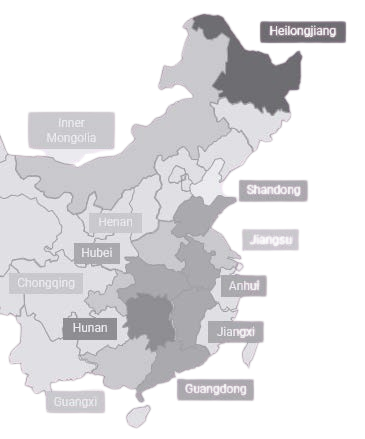
Following registration in Macau or Hong Kong, eligible medicines and devices can access a growing network of hospitals across the Guangdong-Hong Kong-Macau Greater Bay Area, Hainan and a other medical pilot zones in Mainland China. This expanded footprint helps bring treatment to a broad patient population more quickly, while also creating a pathway for patients from across Mainland China to travel to designated hospitals for critical care.
Critical Care Solutions
Pediatric and Clinically Urgent Medications:
- Following registration in Macau, eligible medicines can gain access to leading hospitals in Beijing, Shanghai, Tianjin, Hainan, and Chongqing, as well as 44 of China’s top pediatric hospitals. Classified as clinically urgent and irreplaceable, these medications benefit from a streamlined approval process, entering leading hospitals without the need for national registration or clinical trials.
Fast-Track National Approval:
- Leveraging initial real-world data collected after Macau registration, national registration is typically completed within 3-6 months for Pediatric Orphan Drugs and 6-12 months for Clinically Urgent Medicines. This streamlined process ensures timely access to critical treatments for patients in need.
Major Media Coverage:
- Strategic media outreach and market education can help ensure physicians, patients, and families are aware of available treatment options, supporting timely access for those in urgent need.
network
reach
approval
Asia Market Entry Specialists
On-the-ground execution, broader reach
We support rare-disease programs across therapeutic areas, from early access to multi-country expansion beyond China